FDA allows Houston cancer doctor to resume drug trial
Por um escritor misterioso
Descrição
Federal regulators have lifted a partial hold on a clinical trial performed by Stanislaw

Dabrafenib plus trametinib in patients with BRAFV600E-mutated biliary tract cancer (ROAR): a phase 2, open-label, single-arm, multicentre basket trial - The Lancet Oncology

Texas judges clear Houston cancer doctor of most medical misconduct charges

Early drug development in solid tumours: analysis of National Cancer Institute-sponsored phase 1 trials - The Lancet

Doctor claims to cure pediatric cancer, critics skeptical

Ashish M. Kamat MD Anderson Cancer Center

Drug factory' implants eliminate ovarian, colorectal cancer in mice, Rice News, News and Media Relations

Stephen Hahn - Wikipedia

Gene Editing for Cancer Is Coming of Age, Article

Hagop M. Kantarjian MD Anderson Cancer Center

FDA issues warning to controversial Houston cancer doctor
In the compelling follow-up to the internationally award-winning documentary, Burzynski, the Movie; Burzynski: Cancer is Serious Business, Part II

Burzynski: Cancer Is Serious Business Part II
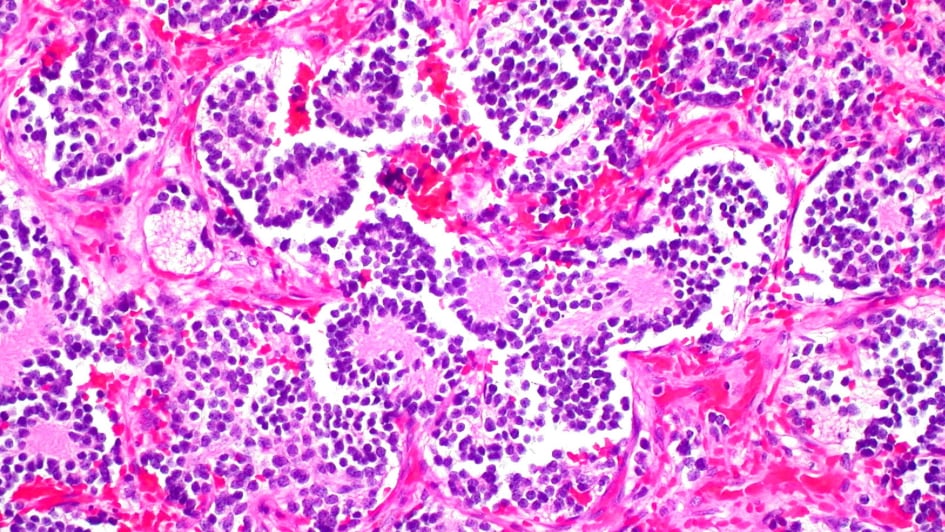
New drug targeting high-risk children's cancer is ready for trials - The Institute of Cancer Research, London

Texas Medical Board sanctions controversial cancer doctor Burzynski

Doctor claims to cure pediatric cancer, critics skeptical
de
por adulto (o preço varia de acordo com o tamanho do grupo)